EXERCISE NO. 4 ANALYSIS OF ASH
I. OBJECTIVES
· To illustrate the use of a muffle furnace.
· To introduce some chemical concepts involved in the ignition of the sample.
II. RESULTS AND CALCULATIONS
In this experiment, the percentage amount of ash present in the three samples after it undergone ignition test being subjected to muffle furnace was studied.
In analytical chemistry, ashing is the process of mineralization for preconcentration of trace substances prior to chemical analysis. Ash is the name given to all non-aqueous residue that remains after a sample is burned, which consists mostly of metal oxides.
Ash is one of the components in the proximate analysis of biological materials, consisting mainly of salty, inorganic constituents. It includes metal salts which are important for processes requiring ions such as Na+ (Sodium), K+ (Potassium), and Ca2+ (Calcium). It also includes trace minerals which are required for unique molecules, such as chlorophyll and hemoglobin.
The experiment began by weighing the crucible and a sample of sky flakes to approximately 0.1 mg. After correct weights were done, the crucible was placed in the muffle furnace for about 650oC until the particles of the sample turned into light gray ash. After observing the presence of gray ash, it was then cooled in the desiccator at room temperature and weighed using analytical balance. The standard values were then gathered after weighing it three times.
The ash obtained differs in composition from the inorganic constituents present in the original biscuits. Incineration causes an expulsion of all water, the loss of carbon dioxide from carbonates, the conversion of iron pyrites into ferric oxide, and other chemical reactions. Ash, as determined by this test method, will differ in amount from ash produced in furnace operations and other firing systems because incineration conditions influence the chemistry and amount of the ash.
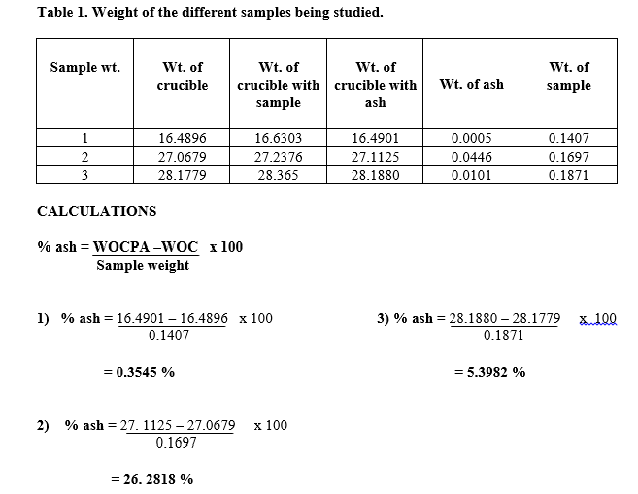
The percentage presence of ash was then calculated using the formula, where WOCPA is the weight of crucible plus ash and WOC is the weight of the crucible. As calculated, sample 1 resulted to 0.3554 % , sample 2 resulted to 26.2818 % and sample 3 was 5.3982%.
Based on the results sited above, the weight of the crucible and the sample being ignited using the muffle furnace forming the ash resulted a decreased in weight. This implies that while the sample was ignited in the muffle furnace, which means the crucible containing the samples was subjected to a very hot environment, the effect of heat on the samples decomposes and produces oxygen. The samples decreased by its mass because the water vapor was loosed producing a dark purple or black crystals.
IV. CONCLUSION
Therefore, based on the experiment being conducted, it can be concluded that a sample being subjected to heat using a muffle furnace which undergone chemical reaction, were the molecules of the substance break apart and join together with those of another substance to create a compound which is a combination of different molecules producing a dark purple or black crystals which is called the ash. Calculating and weighing the samples using a analytical balance before and after the changes occurred, it can be observed that the weights decreased after it was subjected to heat and cooled down. Thus, proper weighing of the samples using an analytical balance is very important way in getting the standard values of the different samples used to analyze ash as a compound that remains after a scientific sample was burned.
V. ANSWER TO QUESTION
1. Differentiate dry ashing from wet ashing process.
ANSWER :
To differentiate this two, dry ashing is usually performed by placing the sample in an open inert vessel and destroying the combustible (organic) portion of the sample by thermal decomposition using a muffle furnace while wet Ashing is treatment of the sample with a moderate amount of sulfuric acid before charring. Charring is performed using an open flame. Liquid samples tend to foam. After the excess sulfuric acid is driven off, the sample is muffled as above.
VI. REFERENCES
· Christian, G.D. 1980. Analytical Chemistry. Third Edition. John Wiley and Sons, New York.
· Fischer, R.B. and D.Y. Peters. 1968. Quantitative Chemical Analysis. W.B. Saunders Co., Philadelphia.
· http://www.inorganicventures.com/ashing-sample-preparation-procedures. Visited on November 9,2015 at 10:00 PM.
· http://www.mikecurtis.org.uk/heating.htm. Visited on November 8, 2015 at 3:00 AM.

 RSS Feed
RSS Feed
